|
If the surface area of the material considered is larger, then there will be a greater number of thermions emitted. Higher is the temperature of the substance, greater is the rate of thermionic emission. However, the energy which must be supplied differs from element to element and is regarded to be its threshold energy or work function. This is because these are the electrons which can be easily freed from the force of attraction so as to enable conduction. When we speak of thermionic emission, our particular interest is in the valence electrons (electrons in the outermost shell). 
by the distribution of electrons surrounding its nucleus. In general, every element can be characterized by its electronic configuration i.e. The number of thermions emitted per second from a substance is known as the rate of thermionic emission. 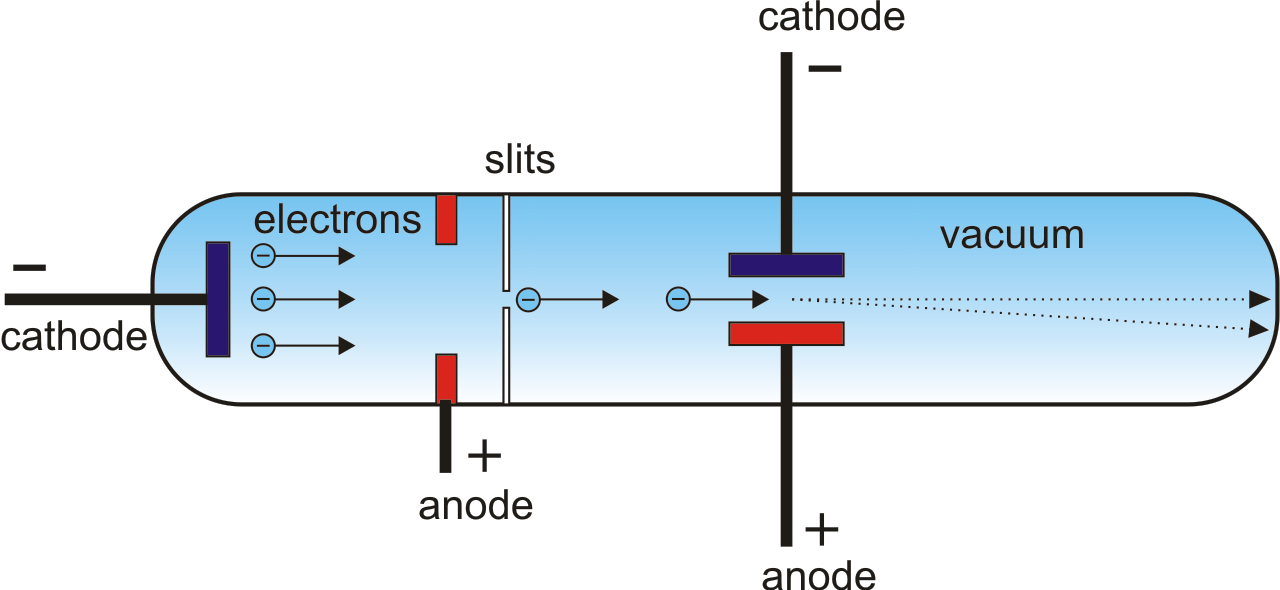
The fact is that the number of thermions emitted is limited due to the effect of space charge – a phenomenon wherein the liberated thermions surround the electrode forming a shield, preventing the emission of further thermions. This causes them to overcome the force of attraction which exists between them and the protons within the respective nuclei.įrom the discussion presented, it might appear that the number of thermions emitted can be increased up to a large value just by increasing the temperature of the substance in-hand. The thermal energy so supplied increases the kinetic energy of the electrons within the material. Now, imagine that we start heating a particular material. These electrons are distributed at various levels around the nucleus and thus possess different levels of energy. At average room temperature, the energy possessed by free electrons in a metal is insufficient to initiate thermionic emission.Īll materials are composed of atoms which in turn consists of a nucleus, made of protons and neutrons, surrounded by electrons. This occurs because the thermal energy given to the carrier overcomes the work function of the material. When a metal is heated sufficiently, the thermal energy supplied to the free electrons causes the emission of electrons from the metal surface. Thermionic emission is defined as the electron emission caused by a sufficiently high level of thermal energy. Indirectly Heated Cathode What is Thermionic Emission?. 
Construction of Cathode for Thermionic Emission.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
